Product Studied :
VigRX Plus®
Year
2012
Published in :
BMC Complementary and Alternative Medicine 2012, 12:155
Methodology :
75 men between 25 and 50 were randomly assigned to take either VigRX Plus or a placebo. Each man had mild to moderate erectile dysfunction as defined by the International Index of Erectile Function (IIEF). Each took two capsules twice daily of either VigRX Plus or the placebo for 12 weeks.
Notable Findings :
Subjects who took VigRX Plus® saw their IIEF erectile function scores rise significantly compared to the placebo after 12 weeks, with an average baseline score from 16.08 (2.87) to 25.08 (4.56) at the conclusion of the study. Similar results were seen in each of the four remaining IIEF domains in orgasmic function, sexual desire, intercourse satisfaction and overall satisfaction.
The investigator’s global assessment rated VigRX Plus as “very good” to “excellent” in more than 50% of participants, compared to “fair” to “good” in about 25% of those who took the placebo.
VigRX Plus® was well-tolerated and more effective than the placebo for improving sexual function in men.
VigRX Scientific Studies Results & Expert Analysis
Research Analysis by Dr. Henry S. Edelson MD, New York University School of Medicine

“At your request, I have performed a review of a product called VigRX. VigRX is offered to the public as an herbal sexual enhancer. Claims related to the product relate to improved sexual function, overall greater satisfaction, and the ability to have and maintain a larger erection than normally experienced by the individual.
I have reviewed all of the ingredients listed on the supplement facts table, and I will address each one and provide a summary at the conclusion of this report. I am also attaching relevant material relating to each component, based on information from a variety of sources, as well as a full MEDLINE search for articles appearing in the medical literature. I will address each ingredient in the order they appear on the supplement sheet.
It is clear to me that the formulation of VigRX has a high likelihood of achieving its stated goal of improving sexual performance, enhancing penile size when aroused, and creating greater pleasure from the sexual act.”
VigRX Clinical Test Results 1

Two dosages of VigRX based on the human dosage used, 15 and 30 mg/kg/D were investigated by orally feeding them to adult male Sprague Dawley rats for 14 consecutive days compared with the control group receiving 1 ml. of distilled water/D.
The results were found that the bodyweight gained from the VigRX-treated groups did not significantly differ from the control group. VigRX, especially at the dosage of 30 mg/kg/D, significantly increased sex drive, erect penile size, intracavernous pressure (ICP) and sperm density in the rats.
Blood testosterone levels of the VigRX-treated group and control group were not statistically different. The body weight and various organs weight, penis, testes, epididymis, seminal vesicle, prostate gland, adrenal gland, spleen and pituitary gland of the VigRX-treated groups did not significantly differ from the control group except that the liver and kidney weight of the VigRX-treated group was significantly less than the control group.
It was concluded that the VigRX at the dosage of 30mg/kg/D could increase the sex drive, penile size, penile erection and sperm density in the rats, whereas VigRX had no effect on the various organ weight. However, it did have some effects on the liver and kidney by decreasing the weight of both organs significantly.
VigRX Clinical Test Results 2
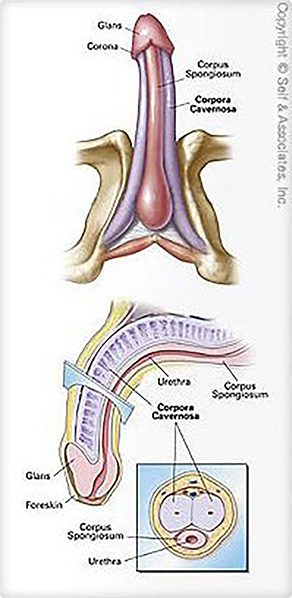
The aim of this project was to investigate the long-term effects of VigRX on sexual behavior at 4, 8 and 12 weeks of treatment. The test studied erect penile size, intracavernous pressure, testosterone levels, sperm density, blood chemistry and some blood parameters, as well as related reproductive, other organ changes after 12 weeks of treatment in adult male Sprague-Dawley rats.
It was found that VigRX at the dosage of 15mg/kg/rat/day fed to male rats for 4, 8 and 12 weeks had no effect on the body weight gained and on sexual behavior of the male rats except ejaculation latency, which was significantly less than the control group.
The long term treatment of VigRX for 12 weeks significantly increased sperm density, the width of erect penile size and intracavernous pressure, but had no effect on every organ’s weight, such as the penis, testes, epididymis, seminal vesicle, prostate gland, liver, kidneys, adrenal gland, spleen and pituitary gland.
Long term treatment of VigRX did not have any effect on blood chemistry value and some blood parameters, except the percentage of lymphocytes. However, the long term treatment of VigRX could significantly increase the testosterone level in these rats.
A histopathological section of some organs, including the liver, kidney and testes revealed that the long term treatment of VigRX had no pathological effects on these three organs.
It was concluded that the long term treatment of VigRX at the dosage of 15 mg/kg/D had no effect on body weight gained and VigRX could not induce the sexual behavior at 4, 8 and 12 weeks of treatment. Long term treatment of VigRX could significantly increase the width of erect penile size, sperm density, intracavernous pressure and testosterone level, but had no effects on organ weight, blood chemistry and some blood parameters in the male rats. The long term treatment of VigRX had no pathological effects on the liver, kidney and testes histology.
VigRX Clinical Test Results Number 3

The aim of this project was to investigate the effects of two dosages of VigRX; i.e., 15 and 30 mg/kg/D fed to male Sprague Dawley rats every day for 14 consecutive days on the liver and kidney of these rats.
It was found that only VigRX at the dosage of 30 mg/kg/D could significantly decrease the liver weight. Histopathological section of both the liver and kidneys showed that both dosages of VigRX did not show any pathological effects on both organs when compared with the control group.
It was concluded that the treatment of VigRX continuously for 14 consecutive days had no pathological effect on the liver and kidney of male Sprague Dawley rats.